Ammonium
Cation, protonated ammonia
Ammonium ▸ Facts ▸ Comments ▸ News ▸ Videos
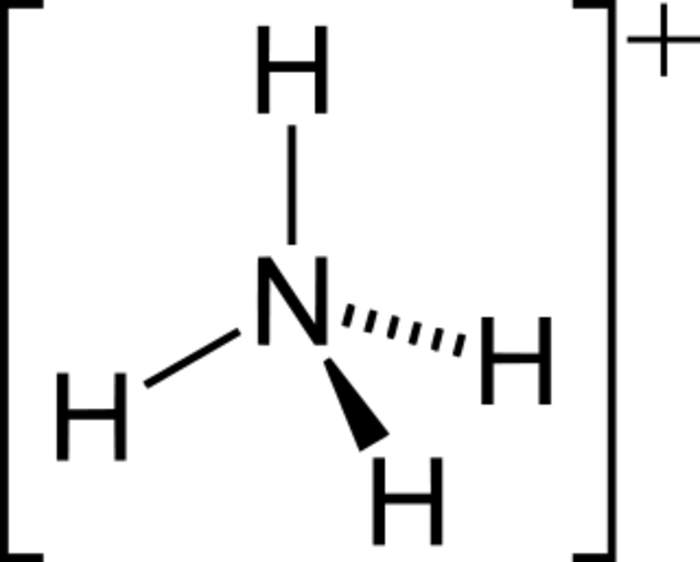
The ammonium cation is a positively charged polyatomic ion with the chemical formula NH+
4. It is formed by the protonation of ammonia (NH3). Ammonium is also a general name for positively charged or protonated substituted amines and quaternary ammonium cations (NR+
4), where one or more hydrogen atoms are replaced by organic groups (indicated by R).
4. It is formed by the protonation of ammonia (NH3). Ammonium is also a general name for positively charged or protonated substituted amines and quaternary ammonium cations (NR+
4), where one or more hydrogen atoms are replaced by organic groups (indicated by R).
| 0 shares | ShareTweetSavePostSend |
You Might Like
No news matches foundSorry, we were unable to find any results in our database for your queryFree news archive accessDid you know? You are eligible to search our news archive with millions of news references free of charge. To do this, please sign in first at the top of the screen. • Information about free access to our news archive Search this site and the web:  |
